Decentralized – also called “virtual” – clinical trials eliminate the complexity of traditional trials’ heavy, multi-site footprint with a central, virtual meta-site which can access patients anywhere, monitor them remotely through tele-health and wearable technologies, and apply modern data science to the larger, more robust data sets that result.
What are Decentralized Clinical Trials?
Traditional vs. Decentralized Clinical Trials
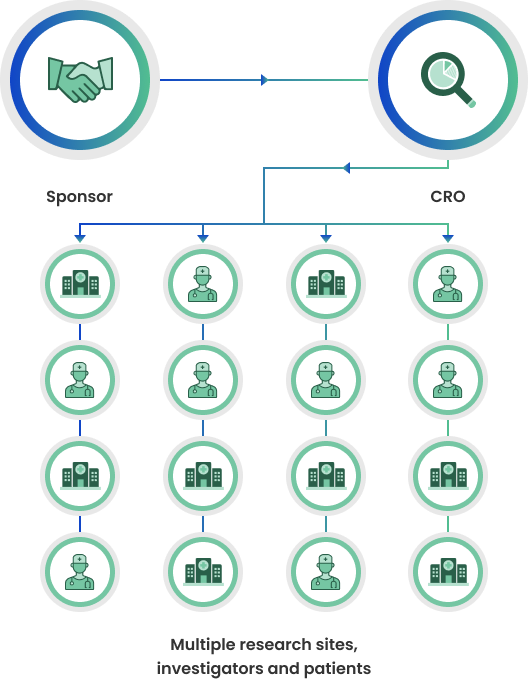
Traditional Clinical Trials
Traditional clinical trials rely on multiple, physical sites to recruit and see patients. Each site has a principal investigator and a study team. While sites are a critical component of many clinical trials, they are also a “choke point” for patient recruitment, requiring patients to be able to physically visit the sites for the duration of the trial.
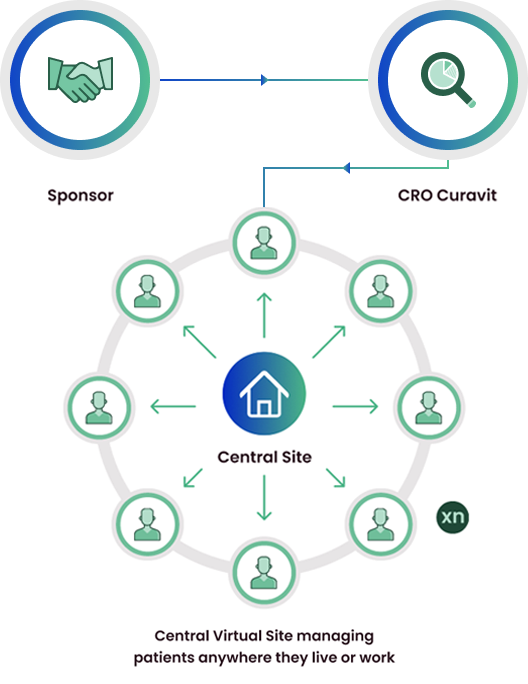
Decentralized Clinical Trials
Decentralized clinical trials replace physical sites with a single, virtual, central site. Patients can be recruited without regard to physical location, and are not required to visit a physical site. Multiple investigators and site teams can be replaced by a single principal investigator and study team. Data are collected via telemedicine, electronic patient reported outcomes, and remote data capture from devices and wearables.

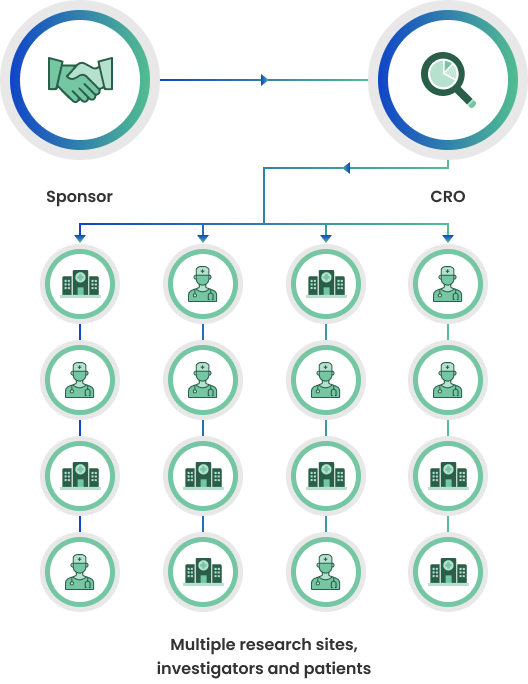
Traditional Clinical Trials
Traditional clinical trials rely on multiple, physical sites to recruit and see patients. Each site has a principal investigator and a study team. While sites are a critical compent of many clinical trials, they are also a “choke point” for patient recruitment, requiring patients to be able to physically visit the sites for the duration of the trial.
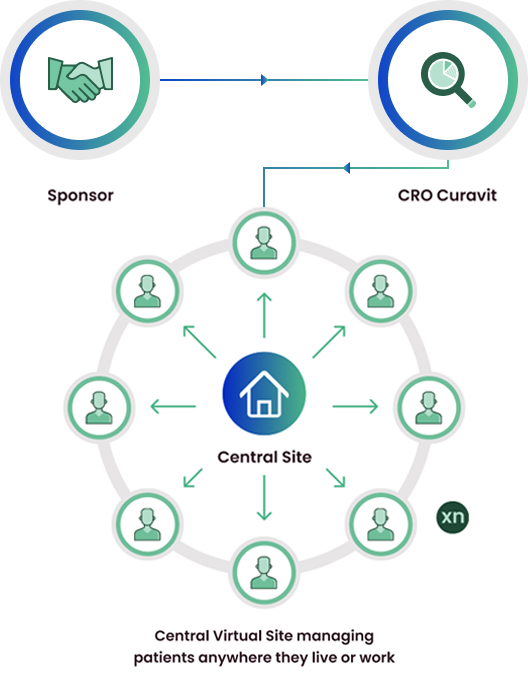
Decentralized Clinical Trials
Decentralized clinical trials replace physical sites with a single, virtual, central site. Patients can be recruited without regard to physical location, and are not required to visit a physicial site. Multiple investigators and site teams can be replaced by a single principle investigator and study team. Data are collected via telemedicine, electronic patient reported outcomes, and remote data capture from devices and wearables.
Learn more about our DCT platform
The Curavit DCT Platform is the digitial manifestation of our decentralized clinical trial services.



